You can Download Metals Questions and Answers, Summary, Activity, Notes, Kerala Syllabus 8th Standard Basic Science Solutions Chapter 7 help you to revise complete Syllabus and score more marks in your examinations.
Kerala State Syllabus 8th Standard Basic Science Solutions Chapter 7 Metals
Metals have some general charac-teristics. They are thermal conductors and electrical conductors. Metals can be beaten into thin sheets by hammering (malleability). Metals can be drawn into thin fine wires (ductility). Hardness is yet another property of metals. The surface newly formed when metals are cut, has a shining appearance (metallic lustre) Metals have the ability to produce sound when tapped with a hard material (sonority).
Metals have high melting point and density. Making use of these characteristics of metals they are used in various purposes. Metals are used to make electric wire because of its electrical conductivity. They are used to make utensils as the metals are thermal conductors. Metals have high density. So they are used to make farming tools. The sonority of metal is utilised to make bells in the places of worship.
There are some similarities in chemical properties of metals. Almost all the metals get tarnished when they are exposed to air for some days. This is because they are react with various components of atmospheric air.
Metals react with water. Metals like sodium, potassium etc reacts with cold water to liberate hydrogen.
Zinc, aluminium, iron, magnesium etc. react with dilute hydrochloric acid to form hydrogen along with salt of metal. There are some metals which does not react with acids.
Ex : Copper.
As the metals react with atmospheric air and water vapour causes corrosion. The iron reacts with air and water vapour. So it undergoes corrosion. Verdigris is formed on copper by reacting with the components of air. Iron and a number of other metals react with different components of air and form new products. This process is known as corrosion of metals. Painting on the metal, electroplating with non corrosive metals are some methods to prevent the corrosion
Metals Textbook Questions and Answers
Kerala Syllabus 8th Standard Chemistry Notes Question 1.
Some metals are listed below. Complete the table by identifying the different uses and the properties which are responsible for the same.
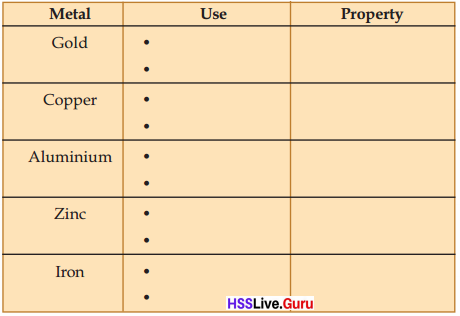
Answer:
| Metal | Use | Property |
| Gold | To make ornaments To make coins | Ductility Malleability |
| Copper | To make utensils To make electric wire | Thermal conductivity Electrical conductivity |
| Aluminium | To make utensils To make aluminium foil | Thermal conductivity Malleability |
| Zinc | To make thin sheets In electric equipments | Malleability Electrical conductivity |
| Iron | To make farming tools To make utensils | Hardness Thermal conductivity |
Hss Live Guru 8 Chemistry Kerala Syllabus Question 2.
Iron is a metal which corrodes fast.
a. What are the factors that favour the corrosion of iron?
b. In coastal regions, copper nails are preferred to iron nails. What could be the reason?
c. Can you suggest some measures to prevent the corrosion of iron?
Answer:
a. Air and water vapour in the atmo sphere
b. Iron rod corrodes quickly in salt water
c. Painting on the metal, electroplating with non corrosive metals
Hsslive Guru Chemistry 8 Kerala Syllabus Question 3.
Based on the physical properties of metals, indicate whether the following statements are true or false
- Aluminium is a conductor of electricity.
- The metal caesium melts at high temperatures.
- Platinum is a metal with poor malleability.
- Potassium is a hard metal.
- Copper, metal is sonorous.
- The density of gold is very low.
- Copper is a good conductor of heat.
- Sodium is a hard metal.
- One of the reasons for the use of gold in making ornaments is its metallic lustre.
- The ductility of tungsten is high.
Answer:
- true
- false
- true
- false
- true
- false
- true
- false
- true
- true
Hsslive Guru 8th Basic Science Kerala Syllabus Question 4.
Which among the metals is stored in kerosene?
(a) Sodium
(b) Iron
(c) Tungsten
(d) Chromium
Why is it stored in kerosene?
Answer:
a. Sodium. Becuase they vigorously react with air.
Kerala Syllabus 8th Standard Physics Notes Question 5.
The names of some metals are given below.
Tungsten, Gold, Sodium, Copper, Iron, Magnesium
Answer the following questions by selecting the appropriate ones from the list.
- Which metal with high malleability is used for making ornaments?
- Which of these metals reacts with cold water?
- Which of these is a hard metal but corrodes easily?
Answer:
- Gold
- Sodium
- Iron
Hsslive Guru Chemistry Class 8 Kerala Syllabus Question 6.
Give reasons for the following statements.
- Tamarind is not stored in aluminium vessels.
- It is a common practice to apply oil on iron articles and tools.
- Stainless steel knives, instead of iron knives, are preferred for cutting citrus fruits.
Answer:
a. Tamarind is acidic. So aluminium corrodes by reacting with acid
b.To reduce the contact with air
c. Gtrus fruit is acidic. It reacts with iron.
Hsslive Class 8 Chemistry Kerala Syllabus Question 7.
Connect a torch bulb to the battery with the different metal wires. Find out which wire gives the highest brightness to the bulb? Arrange the wires in the decreasing order of their electrical conductivity.
Answer:
Iron-copper-aluminium-nichrome
Hsslive.Guru Class 8 Kerala Syllabus Question 8.
Collect the following materials: an iron nail, an aluminium wire, a pencil lead, a copper Beat them hard using a hammer. Which of thenvCan he flattened? What is your conclusion from this experiment?
Answer:
Iron nail, aluminium wire. The metals have malleability.
Class 8 Physics Notes Kerala Syllabus Question 9.
You have understood the properties of metals. Find out the metals mentioned in the following cases.
a. Which metal is used in the storage batteries of vehicles?
b. Most of the metals are solids. Which metal exists as a liquid even at low temperatures?
c. Iron articles are coated with other metals to protect them from corrosion. Which are the two metals usually used for this purpose?
d. Metals have high density. Which is the densest metal?
Answer:
a. zinc
b. mercury
c. zinc, magnesium
d. gold
Metals Additional Questions and Answers
Hsslive 8 Chemistry Kerala Syllabus Question 1.
Tabulate the general properties of metals
Answer:
Hardness, High density, Ductility, Malleability, Sonority, Thermal conductivity, Electrical conductivity.
Hss Live Guru 8 Kerala Syllabus Question 2.
List some of the general properties of metals.
Answer:
Generally metals, are good conductors of electricity, are good conductors of heat, are malleable (can be hammered into thin sheets), are ductile (can be pulled into wires), are solids (except mercury), are hard and strong, have metallic lustre, have high densities, have high melting points, have high boiling points
Hss Live Guru Class 8 Kerala Syllabus Question 3.
What are the situation make use of the following properties of metals Hardness, malleability, thermal conductivity, electrical conductivity, ductility
Answer:
To make farming tools
• To make aluminium foil,
• To make utensils
• To make electrical equipments
• To make ornaments
Hss Live Guru 8th Chemistry Kerala Syllabus Question 4.
Write the following metals in the ascending order of electrical conductivity. Silver, copper, aluminium
Answer:
Aluminium, copper, silver
Hsslive Guru 8th Class Chemistry Kerala Syllabus Question 5.
Even though the electric conductivity is less the wires on electric posts are made up of aluminium. Why?
Answer:
Due to less weight and cost.
Question 6.
Which metal is seen in liquid state?
Answer:
Mercury
Question 7.
Fill the table
| Use | Property |
| To make utensils | ………………………. |
| To make aluminium foil | ………………………. |
| To making farming tools | ………………………. |
| To make electric wire | ………………………. |
| To make bells | ………………………. |
Answer:
| Use | Property |
| To make utensils | thermal conductivity |
| To make aluminium foil | malleability |
| To making farming tools | hardness |
| To make electric wire | electrical conductivity |
| To make bells | sonority |
Question 8.
Butter milk is not kept in aluminium vessels. Why?
Answer:
Butter milk is acid. So it reacts with aluminium
Question 9.
What are the two things which are caused to form verdigris on copper?
Answer:
Air and water vapour
Question 10.
Select the metals which do not react with air.
Copper, iron, gold, magnesium, platinum, sodium, silver
Answer:
Gold, silver, platinum
Question 11.
Write the following rentals in the ascending order of the reactivity with acids. Copper, sodium, iron, aluminium
Answer:
Sodium, iron, aluminium, copper
Question 12.
Why should we paint the iron bars of window?
Answer: To protect them from rusting
Question 13.
What are the components effecting rusting of iron?
Answer:
Air, moisture present in atmosphere
Question 14.
Why sodium and potassium are kept in kerosene?
Answer:
Because they vigorously react with air.
Question 15.
Aluminium vessels get tarnished after few days they purchased. Why?
Answer:
aluminium undergo chemical reaction with air and a covering is formed.
Question 16.
The window bars near sea shore undergo rusting quickly. Why?
Answer:
Iron react with saltwater quickly and undergo corrosion.
Question 17.
Which property of metals is used to make bells.
Answer:
Sonority
Question 18.
What is the property of metal caused for shining appearance?
Answer:
Metallic lustre.
Question 19.
Which is the metal used for making filament of bulb? Which property is made use of it.
Answer:
Tungsten; ductility.
Question 20.
Is there any advantage, if iron nails are kept inside kerosene. Why?
Answer:
Yes, it prevent rusting. Iron nails, when kept inside kerosene, are protected from rusting. They are not in contact with atmospheric air or moisture.
Question 21.
A bottle filled with quicklime is closed with a lid made of iron. After some days, when the lid is opened, which part of the lid is more rusted? What is the reason for that?
Answer:
The outer part of the lid is more rusted. The outer part is in contact with air and moisture; but due to the presence of quicklime, there is no water content inside.
Question 22.
Find the odd one out. Write the reason.
a. Iron, aluminium, sodium, copper
b. Silver, gold, sulphur, lead
Answer:
a. Sodium. Sodium is a soft metal while others are hard metals b. Sulphur. Sulphur is a non-metal others are metals.
Question 23.
Which are the metals found in free state! Why?
Answer:
Metals like gold, platinum, silver, copper etc. are found in free state, . because these metals do not react with air.
Question 24.
To prepare thin sheets, metals are beaten and made into thin sheets.
a. What is the name of this property of metals by which they can be beaten into thin sheets?
b. Which metals can be beaten and made into the thinnest sheet?
Answer:
a. Malleability
b. Gold
Question 25.
Among the statements given below which are true about metals.
a. Metals bum
b. Metals can be beaten into thin sheets.
c. Metals are dull in appearance.
d. Metals are good conductor of heat.
e. Metals conduct electricity.
f. Metals break into small pieces when struck with a hammer.
Answer:
b, d, and e
Question 26.
Does any immediate change occur when a bright nail is exposed to air? What happens to it after a few days?
Answer:
Tmmediate change does not occur. After a few days it reacts slowly with air and gets a coating of oxide.
Question 27.
Why are aluminium rods not used to reinforce concrete?
Answer:
Pure aluminium is thin, not sufficiently hard and strong. So aluminium rods are not used to reinforce concrete.
Question 28.
Iron made objects will undergo quick rusting. Give reason? What are the methods used to prevent the speed of the process?
Answer:
Tron became destroyed when it can react with atmospheric oxygen and moisture form hydrated iron oxide (rust)
- Painting, apply grease or oil
- Covered the iron by using highly active metals like zinc
- Apply tin on iron. ,
- Protect iron made object by applying nickel and chromium on it.