Students can Download Chapter 5 States of Matter Questions and Answers, Plus One Chemistry Chapter Wise Questions and Answers helps you to revise the complete Kerala State Syllabus and score more marks in your examinations.
Kerala Plus One Chemistry Chapter Wise Questions and Answers Chapter 5 States of Matter
Plus One Chemistry States of Matter One Mark Questions and Answers
Question 1.
When a gas is compressed at constant temperature
a) The speeds of the molecules increase
b) The collisions between the molecules increase
c) The speed of the molecules decrease
d) The collisions between the molecules decrease
Answer:
b) The collisions between the molecules increase
Question 2.
The temperature at which a real gas obeys ideal gas law over an appreciable range of pressure is called ___________
Answer:
Boyle temperature or Boyle point
Question 3.
The compressibility factor is given by the expression
![]()
Answer:
a) \(\frac{p V}{n R T}\)
Question 4.
Two flasks of equal volume contain CO2 and SO2 respectively at 298 K and 1.5 atm pressure. Which of the following is equal in them?
a) Masses of the two gases
b) Rates of effusion
c) Number of molecules
d) Molecular structures
Answer:
c) Number of molecules
![]()
Question 5.
With rise in temperature, viscosity of a liquid
a) Increases
b) Decreases
c) Remains constant
d) May increase or decrease
Answer:
b) Decreases
Question 6.
The unit of‘b’ in VanderWaals equation of state.
Answer:
l mol-1
Question 7.
Most probable velocity, average velocity, and root mean square velocity are related by
Answer:
1 : 1.128 : 1.224
Question 8.
The volume of 2.8g of CO at 27°C and 0.821 atm pressure is (R = 0.0821 l atm Km-1 ol-1)
Answer:
3L
Question 9.
The density of gas at 27°C and 1 atm is d. Pressure remaining constant at which of the following temp will its density become 0.75d?
Answer:
400K
Question 10.
The rms velocity of an ideal gas at 27°C is 0.3ms-1. ‘ Its rms velocity at 927°C in (ms-1) is
Answer:
0.6m/s
Plus One Chemistry States of Matter Two Mark Questions and Answers
Question 1.
Find out the relation between the first pair and complete the second pair.
a) Boyle’s law: Temperature
Charles’ law : ……………….
b) Avagadro’slaw: V α n
Ideal gas equation: ……………..
Answer:
a) Pressure
b) pV=nRT
Question 2.
The graphs of Boyle’s law as plotted by Student 1 (Graph 1) and Student 2 (Graph 2) are given below:
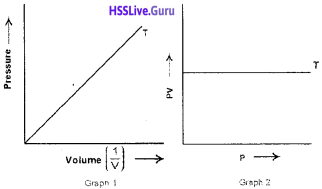
- Which is the correct graph?
- Justify your answer.
Answer:
- Both the graphs are correct.
- According to Boyle’s law, v α 1/p or p α 1/v. This is clear from graph 1. Also, according to Boyle’s law, pv is a constant at constant n and T. This is clear from graph 2.
![]()
Question 3.
The rate of diffusion of hydrogen is less than that of oxygen.
- Do you agree?
- Which law is applied here?
- State the law.
Answer:
- Yes.
- Graham’s law of diffusion.
- Rate of diffusion of a gas is inversely proportional to the square root of its molecular mass.
Question 4.
The ideal gas equation has been modified for real gases by applying pressure and volume corrections.
- What is the corrected equation known as?
- Write the equation and explain the terms.
Answer:
- The van derWaals’equation.
- \(\left(p+\frac{a n^{2}}{V^{2}}\right)\) (V — nb) = nRT
where p – pressure, V – volume, n – no. of moles of the gas, a & b – van der Waals’ constants, R – universal gas constant and T – absolute temperature.
Question 5.
‘Moist soil grains are pulled together.’
- Name the related phenomenon.
- Justify.
Answer:
- Surface tension.
- This is because the surface area of thin film of water in moist soil is reduced due to surface tension.
Question 6.
1. What is aqueous tension?
2. What is its significance in the determination of pressure of a dry gas?
Answer:
1. The pressure exerted by saturated water vapour at a given temperature is called aqueous tension at that temperature.
2. Pressure of dry gas can be calculated by subtracting aqueous tension from the total presssure of the moist gas.
Pdry gas=PTotal-Aqueous tensi0n
Question 7.
A balloon filled with air, when kept in sunlight bursts after some time.
- Name the related law.
- Justify.
Answer:
- Charles’ law
- According to Charles’ law, volume a Temperature. Therefore, the volume increases when temperature increases, When the volume of the gas inside the baloon expanded more than that the balloon could afford, it bursted.
Question 8.
Define surface energy. What is its SI unit?
Answer:
Surface energy is defined as the energy required to rise the surface area of the liquid by one unit. The SI unit of surface energy is J m-2.
Question 9.
a) Based on Boyle’s law how will you show that at a constant temperature, pressure is directly proportional to the density of a fixed mass of the gas?
b) Give the relation between density and molar mass of a gaseous substance.
Answer:
a) According to Boyle’s law,

Question 10.
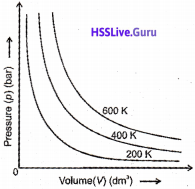
The isotherm of carbon dioxide at various temperatures is given below:

1. What is the significance of the shaded area?
2. Identify the pressure at which liquid CO2 appears for the first time when cooled form 30.98 °C. What is this pressure called?
Answer:
- At any point in the dome shaped shaded area liquid and gaseous CO2 exists in equilibrium.
- 73 atm. Critical pressure (pc).
![]()
Question 11.
Certain properties of liquids are given below: Classify them on the basis of effect of temperature on them,
- Evaporation
- Vapour pressure
- Surface tension
- Viscosity
Answer:
Properties which increase with increase in temperature: Evaporation & Vapour pressure Properties which decrease with increase in temperature: Surface tension & Viscosity
Question 12.
The size of the water bubbles increases on moving to the surface.
1. Name the law responsible for this.
2. What is your justification?
Answer:
1. Boyle’s law.
2. According to Boyle’s law volume is inversely proportional to pressure. At the bottom of the pond, pressure is greater. So the volume (size) of the bubble was the least. But on coming up, pressure decreases and hence size of the bubble increases.
Question 13.
What are the properties of liquid state?
Answer:
Vapour pressure, Boiling point, Viscosity and Surface tension.
Plus One Chemistry States of Matter Three Mark Questions and Answers
Question 1
Analyse the following graph :
1. Name the gas law associated with this graph.
2. State the law.
3. Give the mathematical expression of this law.
Answer:
1. Boyle’s law.
2. It states that at constant temperature, pressure of a fixed amount of gas varies inversly with its volume.
3. Mathematically, Boyle’s law can be written as p ∝ \(\frac{1}{V}\) (at constant T and n)
Or p = k × \(\frac{1}{V}\) where k is the proportionality
constant which depends upon the amount of the gas, temperature of the gas and the units in which p and V are expressed.
![]()
Question 2.
1. What are van der Waals’ forces?
2. Which are the different types of van der Waals’ forces?
3. Arrange the van der Waal’s forces in the increasing order of their strength.
Answer:
1. The attractive intermolecular forces are known as van der Waals’ forces.
2. Dispersion forces/London forces, Dipole-Dipole forces, Dipole-Induced dipole forces and Hydrogen bonding.
3. Dispersion forces/London forces < Dipole-Induced dipole forces < Dipole-Dipole forces < Hydrogen bonding
Question 3.
A graphical representation of Charles’ law is given below:
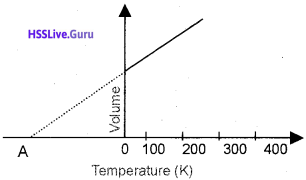
- What is the temperature corresponding to the point‘A’called? .
- What will be the temperature at that point A’ in degree Celsius?
- What is the significance of this temperature?
Answer:
- Absolute zero temperature
- -273.15 °C
- Absolute zero is the lowest hypothetical or imaginary temperature at which gases are supposed to occupy zero volume.
Question 4.
Assume that two gases X and Y at the same temperature and pressure have the same volume.
1. Which of the following is correct?
No. of moles of X= No.of moles of Y
No. of moles of X ≠ No.of moles of Y
2. Which law helped you to find the answer?
3. State the law.
Answer:
1. No. of moles of X= No.of moles of Y
2. Avogadro’s law
3. Equal volumes of all gases under the same conditions of temperature and pressure contain equal number of molecules.
Question 5.
During a seminar session in the class, the presenter argued that equal amounts of both H2and N2 on heating at constant pressure will expand in the same rate. Another student objected this argument by saying that they will expand differently since their molecular masses are different.
- Who is correct in your opinion?
- Which law helped you to reach the answer?
- State the law and give its mathematical expression.
Answer:
1. The argument of the presenter is correct.
2. Charles’ law
3. Charles’ law states that pressure remaining constant, the volume of a fixed mass of a gas is directly proportional to its absolute temperature.
Mathematically, V ∝ T (at constant n and P) Or \(\frac{V}{T}\) = K, where K is the proportionality constant which depends on the pressure of the gas, its amount and the unit in which V is expressed.
![]()
Question 6.
1. What is an ideal gas?
2. Give the ideal gas equation and explain the terms.
3. Derive the ideal gas equation.
Answer:
1. A gas that follows Boyle’s law, Charles’ law and Avogadro law strictly at all conditions is called an ideal gas.
2. The ideal gas equation is pV = nRT where p = pressure, V = volume, n = number of moles, R = universal gas constant and T = absolute temperature.
3. According to Boyle’s law:
V ∝ \(\frac{1}{p}\) at constant T and n.
According to Charles’ law:
V ∝ T. at constant n and p.
According to Avogadro law,
V ∝ n , at constant p and T Combining the above three equations,
V ∝ \(\frac{nT}{p}\)
⇒ V = R\(\frac{nT}{p}\), where R is the proportionality constant known as universal gas constant.
Or pV = nRT, the ideal gas equation.
Question 7.
Partial pressure of a vessel containing Cl2, CO2 and CO is the sum of the partial pressures of Cl2, O2 and CO.
1. If so, is it correct to say partial pressure of a vessel containing NH3 and HCl gases is the sum of their partial
pressures? Justify.
2. Which law helped you to answer this?
3. State the law.
Answer:
1. No. NH3 reacts with HCl to form NH4CI. Since they are not non-interacting gases, their sum of partial pressures may not be equal to the total pressure.
2. Dalton’s law of partial pressures.
3. It states that the total pressure exerted by the mixture of non-reactive gases is equal to the sum of the partial pressures of individual gases.
Question 8.
The average kinetic kenergy of the gas molecules is directly proportional to the absolute temperature.
- Which theory is related to this assumption?
- Write the other postulates of this theory.
Answer:
1. Kinetic moleculartheory of gases.
2.
- The volume of a gas molecule is negligible when compared to the whole volume of the gas.
- There is no force of attraction between the particles of a gas at ordinary temperature and pressure.
- The gas molecules are in random motion.
- During motion, they collide with each other and also with the walls of the container.
- Gravity has no influence in the movement of gas molecules.
- Pressure of a gas is due to the collision of gas molecules with the walls of the container.
- At any particular time, different particles in the gas have different speeds and hence different kinetic energies.
Question 9.
Three mateorological baloons filled with equal amount of helium, rising in the atmosphere are shown below: (Assume that temperature remains constant).
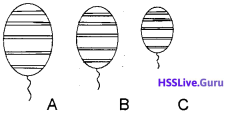
- Which of the baloons will be at the lowest altitude?
- Which law helped you to find the answer?
- State the law.
Answer:
- C –
- Boyle’s law.
- At low altitudes pressure is high. According to Boyle’s law for a given mass of a gas, greater the pressure lower is the volume at constant temperature.
Question 10.
‘All the postulates of the kinetic molecular theory of gases are correct.’
- Do you agree with the statement?
- If no, write the wrong postulates of this theory.
- Give justification.
Answer:
1. No.
2.
- Volume of the molecules of a gas is negligibly small in comparison to the space occupied by the gas.
- There is no force of attraction between the molecules of a gas.
3. If assumption i) is correct, the p vs V graph of experimental data (real gas) and that theoritically calculated from Boyle’s law (ideal gas) should coincide. But this never happens.
If assumption ii) is correct, the gas will never liquify. But gases do liquify when cooled and compressed.
Question 11.
Two gases with equal molecular mass will have the same rate of diffusion.’
- Do you agree?
- Explain.
- Substantiate your answer with an example.
Answer:
1. Yes.
2. According to Graham’s law of diffusion the rate of diffusion depends only on the molecular mass. So if the molecular masses are the same, their rate of diffusion is same.
3. Both CO and N2 have the same molecular mass (28 g mol-1)
Rate of diffusion of CO = Rate of diffusion of N2
![]()
Question 12.
Water can be boiled more quickly on the top of a mountain.
- Do you agree?
- What is the reason?
- What is called boiling point of a liquid?
- How normal boiling point and standard boiling point differ?
Answer:
1. Yes.
2. As we move to the top of a mountain atmospheric pressure decreases and hence boiling point decreases. So water boils quickly.
3. Boiling point of a liquid is the temperature at which the vapour pressure of a liquid is equal to the external pressure or atmospheric pressure.
4. The boiling point at 1 atm pressure is called normal boiling point. The boiling point at 1 bar pressure is called standard boiling point.
Question 13.
Ethanol flows faster than honey.
- Name the related phenomenon.
- Explain this phenomenon.
- What is the effect of temperature on this?
Answer:
1. Viscosity.
2. Viscosity is a measure of resistance to flow which arises due to the internal friction between layers of fluid as they slip past one another while liquid flows.
3. Viscosity of liquids decreases as the temperature rises because at high temperature molecules have high kinetic energy and can overcome the intermolecular forces to slip past one another between the layers.
Question 14.
Liquid drops attain spherical shape.
- Which property of liquids is responsible for this?
- Explain the phenomenon and justify.
- Suggest another consequence of this phenomenon.
Answer:
1. Surface tension.
2. Surface tension is defined as the force acting per unit length perpendicular to the line drawn on the surface of liquid. The lowest energy state of the liquid will be when surface area is minimum. Spherical shape satisfies this condition.
3. Fire polishing of glass – On heating, the glass melts and the surface of the liquid tends to take the rounded shape at the edges due to surface tension, which makes the edges smooth.
Question 15.
Vapour pressure is an important property of liquids.
- What is vapour pressure?
- How boiling point and vapour pressure are related?
- Pressure cooker is used for cooking food at higher altitudes. Give reason.
Answer:
1. Vapour pressure of a liquid is the pressure exerted by the vapour which is in equilibrium with liquid at a given temperature.
2. Boiling point of a liquid is the temperature at which the vapour pressure of liquid becomes equal to the atmospheric pressure. Thus, lower the vapour pressure of a liquid higher will be its boiling point and vice-versa.
3. At high altitudes atmospheric pressure is low. Therefore, liquids at high altitudes boil at lower temperatures in comparison to that at sea level. In a pressure cooker, the internal pressure is greater than atmospheric pressure. Hence, in a pressure cooker water boils at a temperature higher than its normal boiling point of 100 °C. Thus, cooking becomes more effective.
Question 16.
Assume that ‘A’, ‘B’ and ‘C’ are three non-reacting gases kept in a vessel at a constant temperature.
Then, PTotal= PA + PB + PC
1. Name the related law.
2. How can you explain the above law on the basis of kinetic molecular theory of gases?
Answer:
1. Dalton’s law of partial pressures.
2. In the absence of attractive forces, the particles of the gas behave independent of one another. The same is true even if there are more than one type of molecules. Thus, the number of molecules colloding the unit area of the wall per second at a given temperature, fora fixed amount of the gas issame.lt implies that the partial pressure of the gas will be unaffected by the presence of the molecules of other gases. But, the total pressure exerted is duet the impact of molecules of all the gases. Hence, the total pressure would be the sum of the partial pressures of the gases.
Question 17.
1. Write the general equation which relates the different variables of a gas used to describe the state of any ideal gas.
2. A flask at 295 K contains a gaseous mixture of N2 and O2 at a total pressure of 1.8 atm. If 0.2 moels of N2 and 0.6 moles of O2 are present, find the partial pressures of N2 and O2.
3. What is meant by Boyle temperature or Boyle point?
Answer:
1. PV = nRT
2.

3. It is the temperature at which a real gas obeys ideal gas law over an appreciable range of pressure.
Question 18.
1. Liquid tries to rise or fall in the capillary. Name the related phenomenon.
2. What is the effect of temperature on the above phenomenon?
3. WhatistheSI unit of the above phenomenon.
Answer:
1. Surface tension.
2. The magnitude of surface tension of a liquid depends on the attractive forces between the molecules. When the attractive forces are large, the surface tension is large. Increase in temperature increases the kinetic energy of the molecules and effectiveness of intermolecular attraction decreases, so surface tension decreases as the temperature is raised.
3. N m-1.
Question 19.
1. Define critical temperature (Tc ).
2. CO2 cannot be liquified above 31.1°C. Why?
3. The critical temperatures of ammonia and carbon dioxide are 405.5 Kand 304.10 K respectively. On cooling, which of these gases will liquify first? Justify.
Answer:
1. It is the highest temperature at which a gas can
be liquified by applying external pressure.
2. The critical temperature (Tc ) of CO2 is 30.98°C. This is the highest temperature at which liquid CO2 is observed. Above this temperature it is gas.
3. Ammonia. This is because, on cooling, critical temperature of ammonia will be reached first. Liquefaction of carbon dioxide will require more cooling.
![]()
Question 20.
a) Will water boils at higher temperature at sea level or at top of a mountain. Explain.
b) A vessel of 120 mL capacity contains a certain amount of gas at 35 °C and 1.2 bar pressure. The gas is transferred to another vessel of volume 180 mL at 35 °C. What would be its pressure?
Answer:
a) When atmospheric pressure decreases boiling point of the liquid also decreases. So the boiling point of water at sea level is not same as that at the top of a mountain. Atmospheric pressure decreases from sea level as we go high. Hence, the boiling point at the top of the mountain is less than that at sea level.

Question 21.
Real gases deviate from ideal behaviour.
1. What are the two wrong postulates of kinetic theory of gases, responsible for deviation of real gases from ideal behaviour?.
2. When do real gases deviate from ideal behaviour?
Answer:
1.
- Volume of the molecules of a gas is negligibly small in comparison to the space occupied by the gas.
- There is no force of attraction between the molecules of a gas.
2. Real gases deviate from ideal behaviour at high pressure and low temperature, when the gas molecules are very close to each other.
Question 22.
1. What is meant by compressibility factor, Z?
2. What is the significance of compressibility factor?
3. A plot of pV/nRT of oxygen gas against p is as follows:
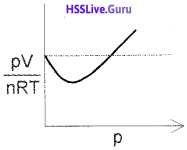
a) Is the gas ideal or real?
b) Write the equation of state of the above gas.
Answer:
1. Compressibility factor (Z) is the ratio of product pV and nRT. Mathematically, Z = \(z=\frac{p V}{n R T}\).
2. The deviation of real gases from ideal behaviour can be measured in terms of compressibility factor.
3. a) Real gas.
\(\left[p+\frac{a n^{2}}{V^{2}}\right]\)[V-nb] = nRT (van der Waals’ equation)
Question 23.
a) What is the difference between gas and vapour?
b) Analyse the vapour pressure vs temperature curve shown below:
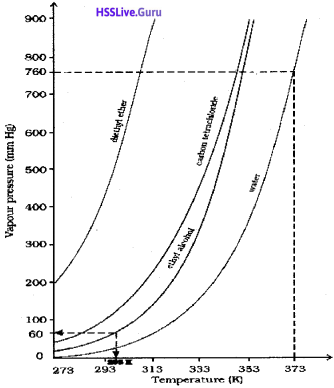
Arrange the compounds shown in the graph in the decreasing order of their normal boiling points,
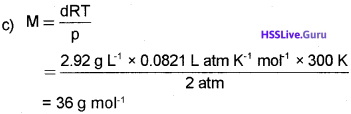
c) The density of a gas was found to be 2.92 g L1 at 27 °C and 2.0 atm. Calculate the molar mass of the gas.
Answer:
a) A gas below its critical temperature can be liquified by applying pressure. Under these conditions, it is called vapour of the substance.
b) water > ethyl alcohol > carbon tetrachloride > diethyl ether
Question 24.
1. How will you account for the observation that automobile tyre is inflated with lesser air in summer than in winter?
2. A sample of gas occupies 250 mLat27 °C. What volume will it occupy at 35 °C if there is no change in pressure?
Answer:
1. This can be explained on the basis of Gay Lussac’s law, according to which at constant volume, pressure of a fixed amount of a gas varies directly with the temperature. In summer season the temperature will be higher. Hence, pressure will increase and the tyre may burst if filled with more air. But during winter temperature is low and hence pressure will below.
2. According to Charles’ law,

Question 25.
Real gases behave ideally at low temperature and high pressure.
- Is the above statement correct or not?
- Justify.
- Write the van der Waals’ equation for 1 mole of a real gas.
Answer:
1. The statement is wrong.
2. This is because real gases behave ideally at high temperature and low pressure, when the gas molecules are far apart.
3. \(\left(p+\frac{a}{V^{2}}\right)\)(V – b) = RT
Question 26.
1. Distinguish between real gas and ideal gas.
2. Explain the deviation of the following gases from ideal behaviouron the basis of the pV vs. p plot. CO, CH4, H2 and He.
Answer:
1. Real gas do not follow, Boyle’s law, Charles law, and Avagadro law perfectly under all conditions. Ideal gas follow, Boyle’s law, Charles’ Law and Avagadro law strictly under all conditions.
2.

It can be seen that at constant temperature pV vs p plots for these gases are not straight lines. Two types of curves are seen. In the curves for H2 and He, as the pressure increases the value of pV also increases. These gases show positive deviation from ideal behaviour at all pressures. The second type of plot is seen in the case of CO and CH4. For these gases the pV value decreases with increase in pressure and reaches to a minimum value characteristics of the gas. After that PV value starts increasing. The curve then crosses the line for ideal gas and after that shows positive deviation continuously.
Question 27.
1. What is meant by laminar flow?
2. Derive the expression for the force responsible for flow of layers of a liquid.
Answer:
1. When a liquid flows overa fixed surface, the layer of molecules in the immediate contact of surface is stationary. The velocity of upper layers increases as the distance of layers from the fixed layer increases. This type of flow in which there is a regular gradation of velocity in passing from one layer to the next is called laminar flow.
2. Consider three layers of a flowing liquid as shown below:
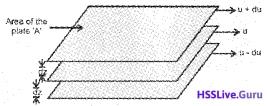
For any layer, the layer above it accelerates its flow and the layer below this retards its flow. If the velocity of the layer at a distance dz is changed by a value du then velocity gradient is given by \(\frac{du}{dz}\). A force is required to maintain the
flow of layers. This force is proportional to the area of contact (A) of layers and velocity gradient.
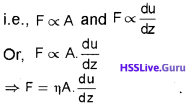
where η is a proportionality constant called coefficient of viscosity.
Question 28.
1. What are London forces?
2. What is the relation between London forces and the distance between the particles?
Answer:
1. The attractive force between two temporary dipoles is known as London forces or Dispersion forces.
2. London forces are always attractive and the interaction energy is inversely proportional to the sixth power of the distance between two interacting particles.
Question 29.
1. What is meant by thermal energy and thermal motion?
2. Can oxygen exist as a gas at -273.15°C? Write the significance of this temperature.
Answer:
1. Thermal energy is the energy of a body arising from motion of its atoms or molecules. The movement of particles due to thermal energy is called thermal motion.
2. At – 273.15 °C oxygen will not exist as a gas. In fact all the gases get liquified before this temperature is reached. It is the absolute zero of temperature, which is the lowest hypothetical or imaginary temperature at which gases are supposed to occupy zero volume.
![]()
Question 30.
Molecues of a gas are in a state of continuous motion
1. What is most probable speed?
2. Give the equation for average speed of molecules.
Answer:
1. Most probable speed is the speed possessed by the maximum fraction of molecules of the gas at a given temperature.
2. If there are ‘n’ molecules in a sample and their individual speeds are u1, u2 ……… un, then, average speed of molecules, uav is given by the equation:
\(u_{a v}=\frac{u_{1}+u_{2}+\ldots+u_{n}}{n}\)
Plus One Chemistry States of Matter Four Mark Questions and Answers
Question 1.
1. State the Avogadro law.
2. Give the mathematical expression of this law.
3. What is the value of molar volume of an ideal gas at 273.15 K and 1 bar?
4. Show that, at constant temperature and pressure, the density of an ideal gas is proportional to its molar volume.
Answer:
1. It states that equal volumes of all gases under the same conditions of temperature and pressure contain equal number of molecules.
2. v ∝ n (at constant P and T)
⇒ V = k × n where k is a proportionality constant.
3. 22.71098 L
4. According to Avogadro law, for n moles of an ideal gas,
V = k × n
But n = \(\frac{m}{M}\) where m is the mass of the gas and M is its molar mass.
Thus, V = k × \(\frac{m}{M}\)
On rearranging the above equation,
M = k × \(\frac{m}{V}\) = k × d
⇒ M ∝ d
Question 2.
The speed of molecules is a measure of their average kinetic energy.
a) What is root mean square speed?
b) Give the equation for root mean square speed.
c) Calculate the following:
i) Root mean square speed of methane molecule at 27°C.
ii) Most probable speed of nitrogen molecule at 25°C
Answer:
a) It is the square root of the mean of the squares of speeds of various molecules of the gas at a given temperature.

Question 3.
The graph A is drawn at high temperature and low pressure and graph B is drawn at low temperature and high pressure.

a) Which graph represents ideal behaviour?
b) Give the equation for combined gas law.
c) A baloon occupies volume of 700 mL at 25°C and 760 mm of pressure. What will be its volume at higher attitude when temperature is 15°C and pressure is 600 mm Hg.
Answer:

Question 4.
1. Give the relationship among the three types of molecular speeds.
2. Drawthe Maxwell-Boltzmann distribution showing all the molecularspeeds.
3. Which of the following molecules will have the higher value of most probable speed at the same temperature, N2 or Cl2? Justify.
Answer:
1. Root mean square speed, average speed and the most probable speed have the following relationship:
Urms > Uav > Ump
2.
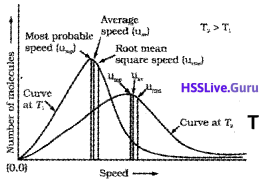
3. N2. This is because at the same temperature, gas molecules with heavier mass have slower speed than lighter gas molecules. At the same temperature, lighter nitrogen molecules move faster than heavier chlorine molecules. Hence, at any given temperature, nitrogen molecules have higher value of most probable speed than the chlorine molecules.
Plus One Chemistry States of Matter NCERT Questions and Answers
Question 1.
What will be the minimum pressure required to compress 500 dm3 of air at 1 bar to 200 dm3 at 30 °C? (2)
Answer:
p1 = 1 bar p2 = ?
V1 = 500 dm³ V2 = 200 dm³
Temperature remains constant.
According to Boyle’s law,

Question 2.
Using the equation of state pV=nRT show that at a given temperature, the density of the gas is proportional to the gas pressure p. (2)
Answer:
According to ideal gas equation :

Question 3.
The density of a gas is found to be 5.46 g/dm³ at 27°C and under 2 bar pressure. What will be its density at STP. (3)
Answer:

Question 4.
Calculate the volume occupied by 8.8 g of CO2 at 31.1°C and 1 bar pressure (R = 0.083 bar L K-1 mol-1). (2)
Answer:
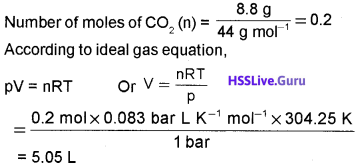
Question 5.
Explain the significance of van der Waal parameters. (2)
Answer:
The van der Waal parameter ‘a’ is a measure of the magnitude of intermolecular forces. The van der Waal parameter ‘b’ which is also called co-volume is a measure of effectve size of the gas molecules.